Freshwater ice can melt into scallops and spikes
- February 15, 2022
- 0
Water’s wacky density leads to strange effects that researchers are still uncovering.
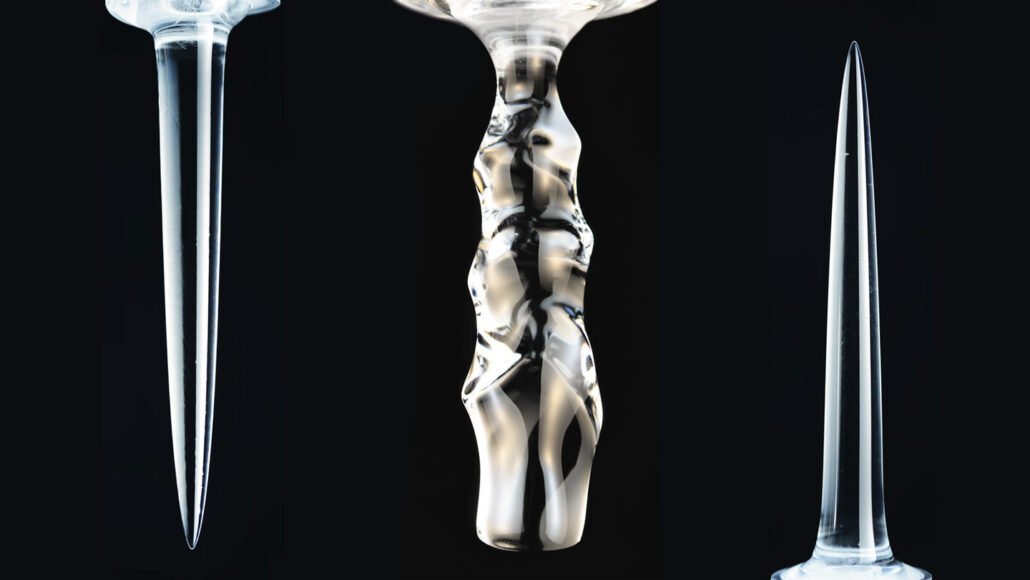
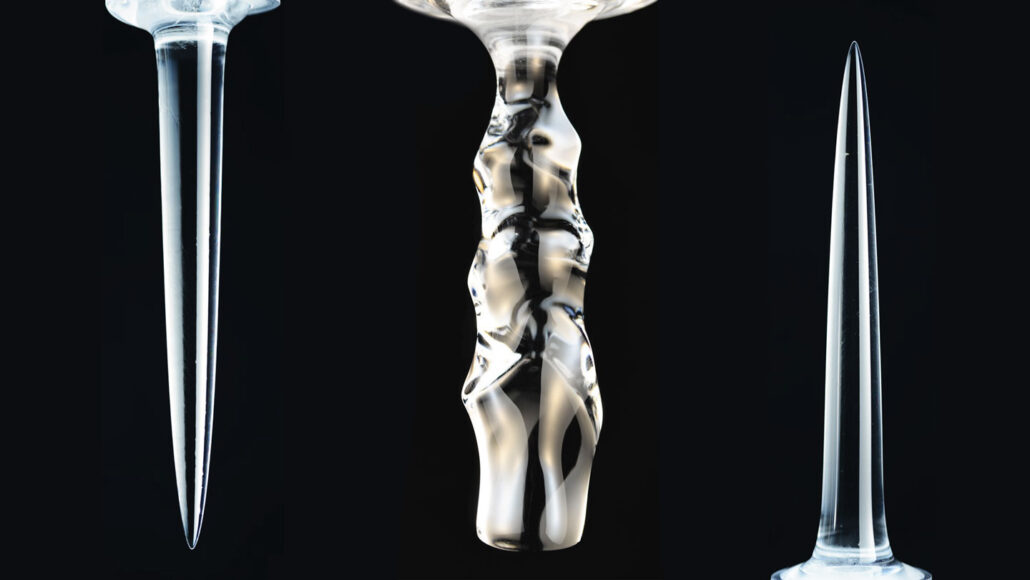
Typically, liquids become denser the more they cool. But freshwater is densest at 4° Celsius. As it cools below that temperature, the water becomes less dense and rises. As a result, ice columns submerged in liquid water can melt into three different shapes, depending on the water’s temperature, researchers report in the Jan. 28 Physical Review Letters.
“Almost everything” about the findings was surprising, says mathematician Leif Ristroph of New York University.
Ristroph and colleagues anchored ultrapure ice cylinders up to 30 centimeters long in place and submerged them in tanks of water at temperatures from 2° to 10° C.
The ice melted into smooth, downward-pointing spikes if placed in water lower than about 5° C. Simulations showed “a strange thing — that the cold liquid water near the ice is actually buoyant” due to being less dense than the rest of the water in the tank, Ristroph says. So that upward flow draws warmer water closer to the ice’s base, causing it to melt faster than the top.
The opposite occurred above about 7° C; the ice formed an upward-pointing spike. That’s because colder water near the ice is denser than the surrounding water and sinks, pulling in warmer water at the top of the ice and causing it to melt faster than the bottom, simulations showed. This matches “what your intuition would expect,” Ristroph says.
Between about 5° to 7° C, the ice melted into scalloped columns. “Basically, the water is confused,” Ristroph says, so it forms different layers, some of which tend to rise and others which tend to sink, depending on their density. Ultimately, the water organizes into “swirls or vortices of fluid that carve the weird ripples into the ice.”
[“source=sciencenews”]